Anodizing aluminum is a process that coats the metal with a protective layer. This layer can be used to improve the appearance of the metal, increase its durability, and protect it from corrosion. This article will discuss how to anodize Aluminum using different methods. We will also discuss the benefits of anodizing Aluminum and how to choose the right color for your project.
The following are the steps for anodizing Aluminum at home
Step 1: Prepare the material.
① Buy some standard aluminum parts. Anodizing works particularly well on aluminum alloys, so it can be done at home with little care. Try to start with small parts so you can drown it with just a little acid.
② Buy a thick plastic tub to soak metal parts in. Choose a very strong and durable plastic material.
③ Buy some clothing dye at your local grocery store. In anodizing, you can stain a metal with almost any color. Apple’s iPods are dyed the same way.
You can also buy anodized dyes for better results.
④ Go to a hardware store and buy some oil remover, two long lead cathodes, and a roll of aluminum wire.
⑤ Get plenty of purified water, baking soda, and rubber gloves.
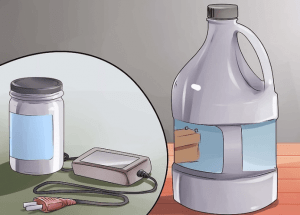
⑥ Find a place to buy a few liters of sulfuric acid (battery acid), lye, and a regulated power supply of at least 20 volts. Battery acid may not be easy to find, but you can probably find it at auto parts stores. You can use a large battery charger as a regulated power source.
Step 2: Clean the aluminum alloy surface
① Wash metal surfaces with soap and water.
② Dip a rag in oil remover to remove grease from the metal surface.
③ Mix 45 ml of lye with 4 liters of pure water. The mixture can be packed in a smaller plastic bowl or an unused metal bowl. Soak the parts in it for 3 minutes, then remove and rinse.
The lye will remove the oxide layer existing on the metal surface. After cleaning up, the metal surface is not easy to hang water and will not leave water beads.
Wear rubber gloves when using lye.
Do not use spoons or measuring cups for food. The materials used in this process are toxic.
Step 3: Prepare the anodizing tank
① Place the plastic basin in a well-ventilated area, away from things that may be damaged during handling. Place a piece of plywood or thick cloth under the plastic basin to prevent liquid spillage.
Keep the temperature in your room between 21 and 22 degrees Celsius for best results.
② Get the power supply ready. Place it on a non-flammable material, such as concrete, and make sure it is always working.
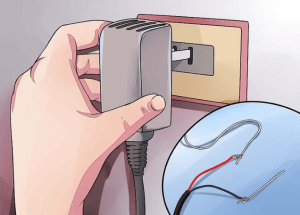
You need to connect the positive terminal of the battery charger or rectifier to the wire on the aluminum alloy part.
You need to connect the negative end of the battery charger to the aluminum wire connected to the 2 lead cathode plates.
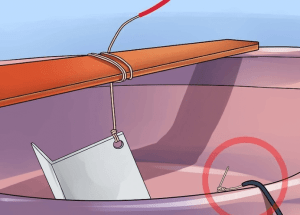
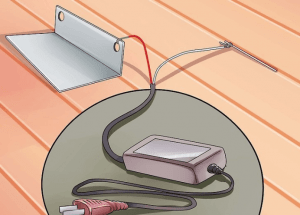
③ Connect one end of the long aluminum wire to the aluminum alloy part. You can use 12 gauge aluminum wire. Wrap or attach it to an inconspicuous part.
The area of the part connected with the wire will not be anodized.
Make sure you wrap it tightly so that the current is more stable.
④ Wrap the wire around a thin piece of wood that sits across the plastic basin so you can easily remove the parts when you’re finished. When wrapping wires, leave enough wires to connect to the power source.
Try the thin wood to ensure the aluminum parts are completely immersed in the acid.
⑤ Place a lead cathode sheet on each side of the basin. The cathode pieces were connected with aluminum wires and then joined on a thin board. You need to connect this wire to the negative end of the power supply.
Ensure the wires connecting the aluminum alloy parts do not contact the lead cathode plate.
⑥ Pour the purified water and battery acid into a large plastic bowl in a one-to-one ratio. The amount used depends on the size of the metal part you are anodizing. Be careful not to let the acid spill.
Be sure to pour the water first and then add the acid.If you accidentally spill acid, quickly sprinkle baking soda to counteract it.
Wear a mask or respirator before you begin treating the acid. Turn on a fan to ventilate the area.
⑦ Connect the aluminum wire connecting the aluminum alloy part to the positive terminal of the power supply. Connect the aluminum wire connecting the lead cathode plate to the negative electrode of the power supply.
⑧ Check to ensure there aren’t any leaks, the power connection is strong, and you don’t have exposed skin.
Step 4: Start anodizing and coloring for Aluminum
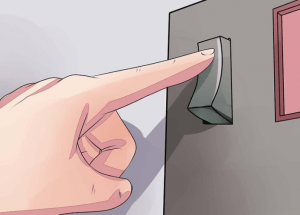
① Turn on the power. Turn up the power slowly. You want to use 12 amperes per square foot of material.
Increasing the power too fast or too high will lead to burning aluminum wire.
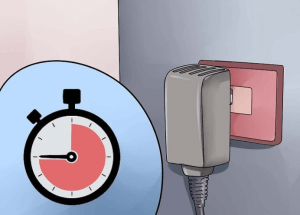
② Let the power supply stabilize for 45 minutes. You will see a small number of oxidation bubbles forming on the aluminum alloy’s surface. And the color will start to turn brown and then yellow.
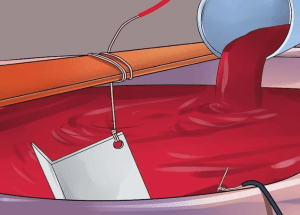
③ Dye is mixed in the anodizing process. The Dye is mixed with purified water and heated to 37 to 60 degrees Celsius.
④ Turn off the power. Remove metal parts and rinse with pure water.
⑤ Place the aluminum alloy parts in a warm dyeing bath. Let sit for 15 minutes.
⑥ Boil pure water in an electric heater. Remove the aluminum alloy parts from the Dye and cook them in boiling water for 30 minutes.
⑦ Carefully remove the hot parts and leave them to dry. At this point, a colored oxide layer should form on the surface of the metal.
All materials and tools
- Aluminium alloy parts
- Work clothes
- Rubber gloves
- goggles
- Thick plastic POTS
- Neutral soap
- water
- Pure water
- Plastic or metal bowl
- In addition to the agent
- lye
- Small lead cathode sheet
- The old measuring spoon
- Baking soda
- Power supply (e.g. car battery charger)
- Breathing apparatus/fan
- The old metal plate
- Electric/gas stove
Anodizing is a simple process that can be used to protect metal from corrosion. It is also a good way to give metal a unique color. With a little practice, you can anodize any metal you want. Try it out on your next project! Thanks for reading.
Try Made by Aria Now
All information and uploads are secure and confidential.
Categories
Share On
Recent Post
What is Overmolding? A Complete Guide
In today’s manufacturing world, creating durable, functional, and aesthetically pleasing
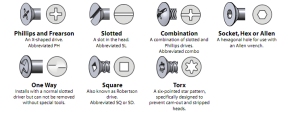
A Complete Guide on the Types Of Screw Head
You’ve probably stood in front of a confusing display of